Xeomin
Xeomin - New generation of botulinum
Xeomin is a product of MERZ drug factories in Germany. It was officially approved by the Canadian government in 2009. It was certified by the US FDA in 2011. The product is also officially named Xeomin;. The product was approved by the Canadian government in 2009. The product was approved by the Canadian government in 2009. The product was certified by the United States in 2011. The product is also officially named Xeomin;.
Xeomin is a kind of high cleanliness botulinum type A botulinum (Botulinum neurotoxin type A), which is characterized by that it does not contain complex protein, even after long-term injection or high dose injection, it will not produce resistance to human body, the effect is good, and the problem of sensitivity is less.
Xeomin Features
- Products approved by the United States FDA, CE, Hong Kong Health Department, the safety products that can be used by adults
- Does not include the compound protein
- Cannot cause the human body to produce the immune body
- Same function with BOTOX”, suits the use in the small scope partial treatment
Injectable location and use
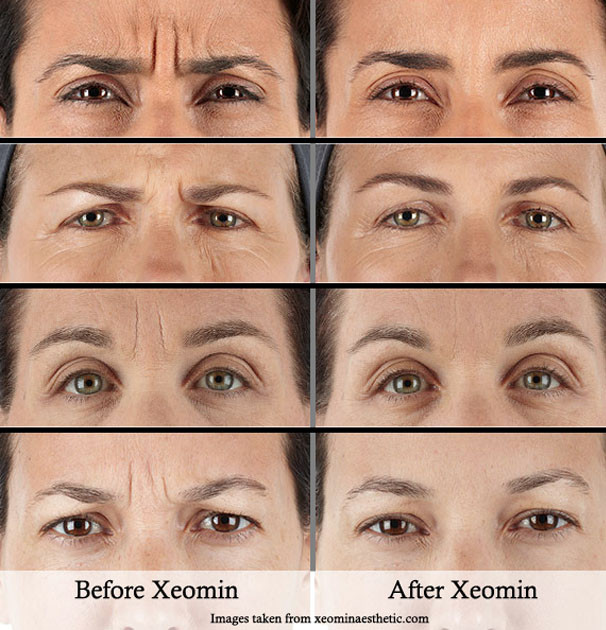
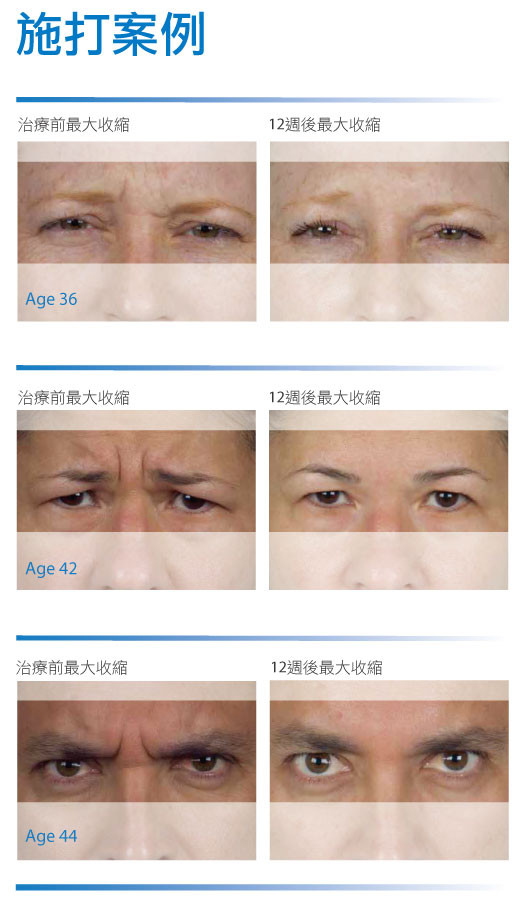
- Fixed dynamic expression lines: forehead wrinkles, crow’s feet, frown lines, and other wrinkles nose.
- Improve face type: masticatory muscle size because each person’s facial contours will be different, there are also some people due to excessive chewing habits make face muscle movement, resulting in the case of asymmetric face.
- Thin legs: because of congenital or acquired excessive amount of exercise, causing the calf muscles of the situation.
- Antiperspirant: excessive sweat secretion for some people is very inconvenient, especially in the summer.